In fields such as industrial pure water preparation, biopharmaceutical production, food and beverage processing, and precision electronics manufacturing, excessive microorganisms in water can affect product quality and the stable operation of equipment. Impurities such as bacteria, mold, and microbial colloids not only cause water turbidity and substandard quality, but also trigger a series of production failures, including biofilm growth in pipelines, membrane element blockage, and product contamination. Therefore, a core question of widespread concern is: Will reverse osmosis remove bacteria? Can the pure water treated by reverse osmosis meet industrial sterile water standards? This article, based on actual operating conditions, provides an in-depth analysis of the sterilization principle, actual effect, application limitations, and supporting optimization solutions of reverse osmosis.
1.The principle of reverse osmosis remove bacteria
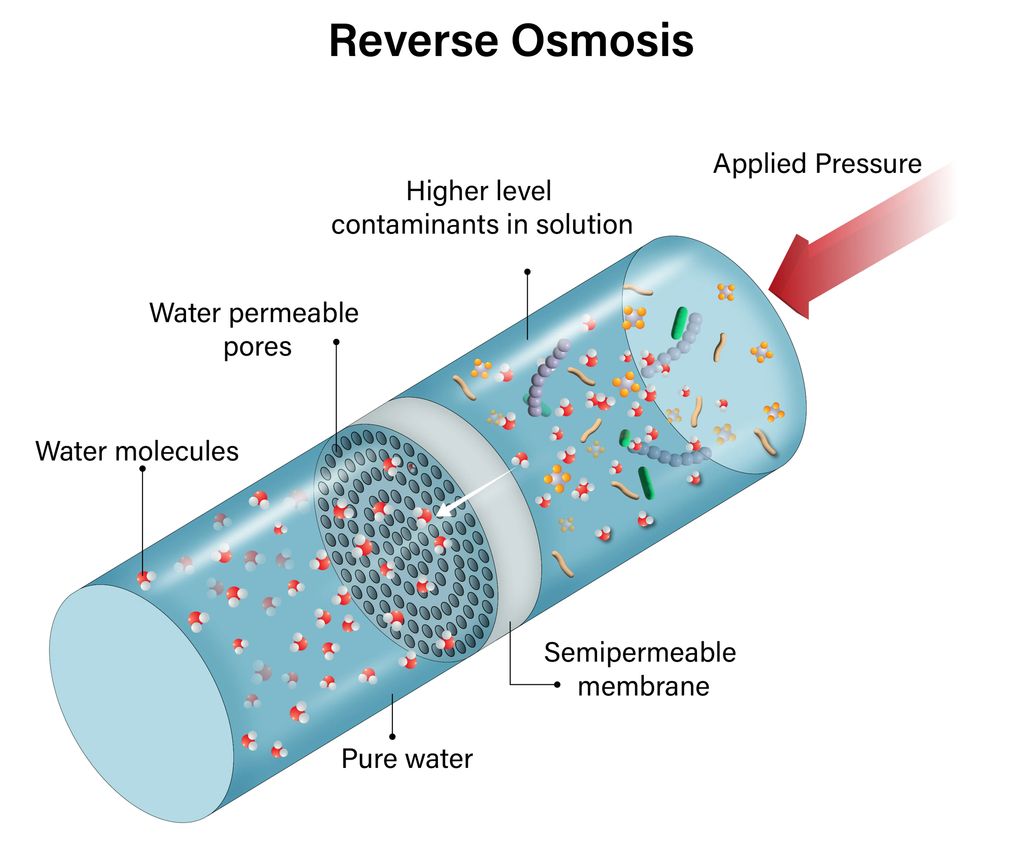
The logic behind reverse osmosis for removing bacteria is a physical sieving and separation process. Unlike chemical or biological sterilization methods such as ultraviolet light, ozone, and disinfectants, this process involves no chemical reactions and leaves no chemical residues, making it suitable for most demanding industrial water usage scenarios.
Reverse osmosis membranes are high-precision semi-permeable membranes. Conventional industrial RO membranes have pore sizes of only 0.1-1 nanometers, while common industrial pathogens and other bacteria in water, such as E. coli, Legionella, and heterotrophic bacteria, typically have cell sizes between 500-5000 nanometers, far larger than the membrane pore size. Therefore, driven by the pressure provided by a high-pressure pump, the raw water overcomes the natural osmotic pressure, allowing water molecules to freely permeate the reverse osmosis membrane. Meanwhile, impurities such as bacterial cells, microbial spores, microbial colloids, and large organic molecules are physically intercepted on the membrane surface and discharged from the system with the concentrated water.
This precise screening mechanism physically blocks the path of bacteria into the water production end, achieving highly efficient retention of bacteria in the water without relying on bactericides. Therefore, will reverse osmosis remove bacteria? The answer is obvious: reverse osmosis possesses stable sterilization capabilities. Furthermore, reverse osmosis membranes can simultaneously intercept viruses, heavy metal ions, suspended solids, and other impurities, achieving comprehensive water purification.
2.Reverse osmosis remove bacteria and have antibacterial effect
The standard configuration of the industrial reverse osmosis system can consistently achieve a bacteria removal rate of over 97%, fully meeting the microbiological requirements for ordinary industrial pure water, process cleaning water, and boiler feedwater. For common suspended and free bacteria in water, the reverse osmosis membrane provides virtually no blind spots, and the total bacterial count in the effluent can be controlled at extremely low levels.
In high-end industrial applications such as biopharmaceuticals and aseptic food processing, the first-stage reverse osmosis system can initially achieve basic aseptic water quality. When combined with subsequent advanced treatment processes (EDI modules), it can meet the standards for pharmaceutical purified water and aseptic process water.
Compared to traditional sand filters, carbon filters, and precision filters, reverse osmosis has an absolute advantage in microbial purification capabilities. Conventional pre-filtration equipment can only intercept large particulate impurities and cannot remove microscopic bacteria, while reverse osmosis can achieve precise filtration at the molecular and microbial levels.
3.Industry limitations of reverse osmosis remove bacteria
Many users mistakenly believe that installing reverse osmosis system guarantees permanent sterile water production without any problems. In reality, RO technology only physically traps bacteria. It cannot kill them. This is its core limitation and a major cause of industrial water treatment failures. The large number of bacteria and microorganisms trapped on the reverse osmosis membrane surface accumulate continuously, multiplying rapidly, especially under suitable water temperature and quality conditions, gradually forming a biofilm.
This biofilm directly clogs the membrane pores, leading to decreased reverse osmosis system permeate flow, increased transmembrane pressure, and reduced desalination rate. This not only deteriorates the quality of the produced water but also significantly shortens the membrane element’s lifespan. More importantly, when the RO system shuts down, experiences pressure fluctuations, or suffers minor membrane damage, the bacteria accumulated on the membrane surface can penetrate the membrane, causing secondary contamination of the produced water’s microorganisms and triggering production accidents. Furthermore, for some ultramicroscopic microbial spores, the RO process alone has limited interception stability and cannot fully meet the extremely stringent requirements for ultrapure water and sterile pharmaceutical water.
4.Optimized solution for reverse osmosis remove bacteria
To compensate for the shortcomings of single reverse osmosis technology in sterilization, the industry generally adopts a combined process of “pretreatment + reverse osmosis + deep sterilization”. The specific process is as follows:
- Implement proper pretreatment protection. Install precision filters and activated carbon filters at the front end of the reverse osmosis system, and regularly add non-oxidizing bactericides to kill most bacteria in the raw water in advance and reduce the accumulation of microorganisms on the membrane surface.
- Secondly, develop a standardized operation and maintenance plan. Regularly perform chemical cleaning and system sterilization of the reverse osmosis membrane to remove biofilm and residual microorganisms from the membrane surface and restore the filtration performance of the membrane elements.
- For high-end industrial applications, a post-treatment deep sterilization process is required. Ultrafiltration, UV sterilizers, or ozone disinfection equipment should be installed downstream of the reverse osmosis product water to create multiple sterilization barriers. Ultrafiltration can further intercept tiny microbial spores, while UV light can kill any remaining free bacteria in the water, completely eliminating the problem of excessive microbial levels.
Summarize
In summary, will reverse osmosis remove bacteria? The answer is yes, reverse osmosis technology can efficiently remove bacteria from water. However, it must be clear that reverse osmosis is “retention filtration,” not “sterilization and inactivation.” A single reverse osmosis process cannot achieve a long-term, stable supply of sterile water and carries the risk of secondary biofouling outbreaks.
In practical applications, we need to combine pre-treatment sterilization, regular system maintenance and cleaning, and post-treatment deep sterilization processes to form a complete water treatment system. Only then can we maximize the sterilization advantages of reverse osmosis and continuously and stably produce sterile industrial water that meets standards. If you need to learn more about sterilization processes, please feel free to contact us for solutions.