Dans le contexte actuel de développement de haute qualité de l'industrie pharmaceutique, le contrôle de la qualité de l'eau est devenu crucial pour la qualité et la sécurité des médicaments. L'eau pharmaceutique n'est pas seulement une matière première importante pour la production de médicaments. Mais sa pureté est également un facteur clé pour la stabilité de l'efficacité des médicaments et la réduction du risque d'effets indésirables.
L'équipement de traitement de l'eau EDI, nouveau type d'équipement remplaçant la technologie traditionnelle d'échange d'ions, devient la solution privilégiée pour préparation d'eau de haute pureté dans l'industrie pharmaceutique en raison de ses principaux avantages, à savoir l'absence de pollution chimique et la stabilité de la production d'eau.
Comment fonctionnent les équipements de traitement de l'eau EDI ?
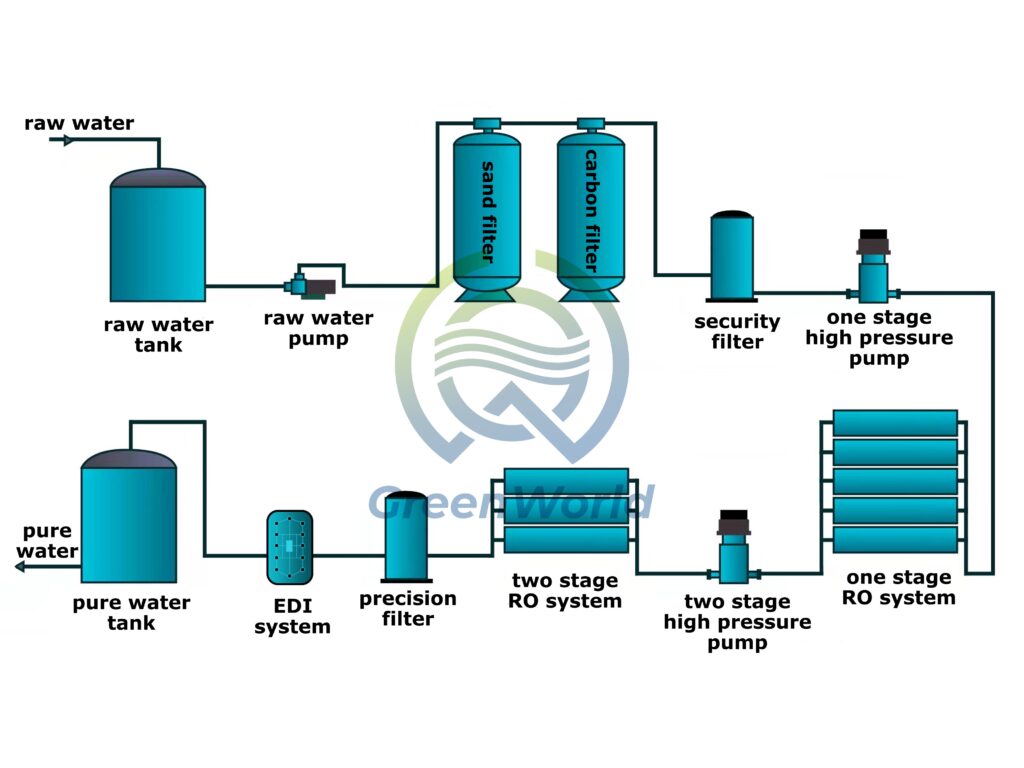
Grâce à sa technologie de pointe, l'équipement de traitement d'eau EDI répond parfaitement aux exigences rigoureuses de l'industrie pharmaceutique. Cet équipement intègre de manière innovante des résines échangeuses d'ions, des membranes échangeuses d'ions sélectives et un champ électrique continu pour former un système de purification d'eau hautement performant.
Compte tenu des caractéristiques spécifiques de l'eau pharmaceutique, l'eau brute doit d'abord subir un processus de prétraitement précis afin d'éliminer complètement les solides en suspension, les colloïdes, les micro-organismes et autres contaminants.
Il entre ensuite dans le module central EDI. À l'intérieur du module, les membranes d'échange de cations et les membranes d'échange d'anions sont disposées en alternance, créant ainsi des zones indépendantes de dessalement et d'évacuation des concentrés.
Sous l'effet d'un champ électrique continu, les ions nocifs présents dans l'eau migrent de manière directionnelle. Par exemple, les cations se déplacent vers la cathode et les anions vers l'anode, ce qui permet une séparation efficace des ions et des molécules d'eau grâce à la perméation sélective de la membrane.
Plus important encore, le champ électrique active en permanence la capacité de régénération de la résine échangeuse d'ions, ce qui élimine complètement le recours aux régénérateurs acido-basiques dans les processus traditionnels et évite le risque de pollution chimique à la source.
Pourquoi la préparation d'eau de haute pureté nécessite-t-elle un équipement de traitement d'eau EDI ?
En ce qui concerne la pureté de l'eau, l'eau ultrapure produite conserve une résistivité stable de 10-18 MΩ-cm, ce qui est parfaitement conforme aux normes rigoureuses applicables à l'eau pour injection et à la production d'API stériles dans le cadre des bonnes pratiques de fabrication (BPF) pour les produits pharmaceutiques. Simultanément, il évite efficacement les effets négatifs des ions d'impureté sur les réactions de synthèse des médicaments et la stabilité de la formulation.
En termes de sécurité, l'équipement EDI ne nécessite aucun ajout de réactifs acides ou alcalins pendant toute la durée de son fonctionnement. Cela permet d'éviter la production d'eaux usées chimiques susceptibles de polluer l'environnement et d'éliminer le risque de contamination croisée dû aux résidus du régénérateur.
Au niveau opérationnel, bien que l'investissement initial en équipement soit relativement élevé, il élimine les coûts ultérieurs tels que l'achat du régénérateur, le traitement des eaux usées et le fonctionnement manuel, réduisant ainsi les coûts d'exploitation à long terme de plus de 30%. Il évite également les problèmes d'interruption de la production d'eau pendant la régénération dans les procédés traditionnels, ce qui garantit la continuité de la production pharmaceutique.
Quelles étapes de l'industrie pharmaceutique utilisent des équipements de traitement de l'eau par EDI ?
Dans l'ensemble de la chaîne de l'industrie pharmaceutique, les équipements de traitement de l'eau EDI (dioxyde électronique) ont pénétré de multiples étapes clés de la production.
① Dans la production de médicaments injectables, ils utilisent l'eau ultrapure stérile générée par notre équipement pour la préparation des solutions médicamenteuses et le nettoyage des conteneurs - mettant à profit 15 ans d'expertise en matière de traitement des eaux industrielles pour garantir directement la stérilité et la stabilité des médicaments injectables et prévenir les réactions pyrogènes causées par des problèmes de qualité de l'eau.
② Dans la synthèse d'ingrédients pharmaceutiques actifs (IPA) stériles, l'eau de haute pureté, en tant que milieu réactionnel, peut améliorer l'efficacité de la réaction et réduire la formation d'impuretés, garantissant ainsi que la pureté de l'IPA répond aux normes.
③ Dans la production d'excipients pharmaceutiques et d'emballages de médicaments, ils utilisent l'eau générée par l'EDI pour la purification des excipients et le nettoyage de précision des matériaux d'emballage.
④ En outre, dans les laboratoires de R&D pharmaceutique, l'eau stable de haute pureté fournie par l'équipement EDI offre une qualité d'eau fiable pour des expériences telles que l'analyse des composants de médicaments et le développement de formes posologiques.
Résumer
À l'avenir, grâce à l'innovation des membranes échangeuses d'ions et des matériaux de résine, l'équipement EDI renforcera encore les capacités de contrôle aseptique tout en améliorant l'efficacité de la production d'eau et en réduisant la consommation d'énergie. Cela permettra de mieux répondre aux besoins spécifiques des secteurs pharmaceutiques haut de gamme tels que les produits biopharmaceutiques et les médicaments issus du génie génétique.
Si vous êtes intéressé par ce type d'équipement, n'hésitez pas à nous contacter. nous contacter.